All Science News
A first glimpse at our Galaxy's magnetic field in 3D
Thanks to new sophisticated techniques and state-of-the-art facilities, Astronomy has entered a new era in which the depth of the sky can finally be accessed.
Of bantam brains and fancy footwork: bioinformatics tools help reveal complexity of avian evolution
In 2014 the Science journal featured an article on the bird tree of life, mentioning the essential role of algorithms and supercomputers that enable modern research in evolutionary biology for all types of living beings. Now, a decade and a giant leap in tool development later, part of the team that coordinated the computer analyses at that time co-authored another paper in Nature on the complexity of avian evolution.
Greek researchers from FORTH, NKUA and Harvard University reveal a new mechanism that regulates intestinal stem cells
In an article published recently at the high-impact journal Nature Communications, researchers from NKUA, FORTH-IMBB, and Harvard University, led by Prof. Aristides Eliopoulos of NKUA and postdoctoral researcher Dr. Zoe Veneti of FORTH-IMBB, reveal a new mechanism that regulates the proliferation and differentiation of intestinal stem cells.
Prestigious ERC Consolidator Grant awarded to FORTH Researcher Dr. Panagiotis N. Moschou
Panagiotis N. Moschou, Researcher at the Institute of Molecular Biology and Biotechnology (IMBB) of the Foundation for Research and Technology – Hellas (FORTH) and Associate Professor at the Department of Biology, University of Crete is awarded a Consolidator Grant from the European Research Council (ERC).
FORTH attracts significant funding through GSRI’s Flagship Project
ERC Starting Grant Awarded at the Institute οf Astrophysics of FORTH to study Violent Phenomena around Supermassive Black Holes
Ioannis Liodakis, a NASA postdoctoral fellow at the Marshall Space Flight Center, was recently awarded the prestigious European Research Council (ERC) Starting Grant.
ERC Starting Grant awarded to a postdoctoral researcher at the Institute of Astrophysics of FORTH for her research on supermassive black holes
It is worth mentioning that FORTH hosts the largest number of ERC Grants (40) in Greece.
Major new funding from the European Research Council to the Institute For Mediterranean Studies of FORTH
Elena Anagnostopoulou, Professor of Linguistics at the University of Crete and Collaborating Faculty Member of the Institute for Mediterranean Studies (IMS) of the Foundation for Research and Technology-Hellas (FORTH), has been selected for funding from the competitive European funding program ERC Advanced Grant 2022.
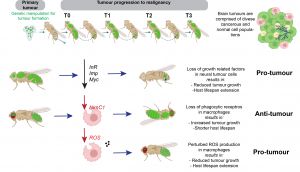
New study reveals the mechanisms that drive neural tumour progression to malignancy
A new research study FORTH-IMBB, published in the international peer reviewed science journal PNAS, reveals how cancer cell intrinsic mechanisms and interactions with the microenvironment shape brain tumour growth and progression to malignancy.
Page:123456789 | Next >