All Science News

Protein structural dynamics underlie their evolution and adaptability over the centuries
How does nature utilize the common repertoire of folds in proteins to differentiate their specificity, and ultimately their function? This long-standing and fundamental question was addressed in a study conducted at the Institute of Molecular Biology and Biotechnology of the Foundation for Research & Technology-Hellas (FORTH-IMBB). The results of this work were recently published in the leading international journal PNAS (Proceedings of the National Academy of Sciences).
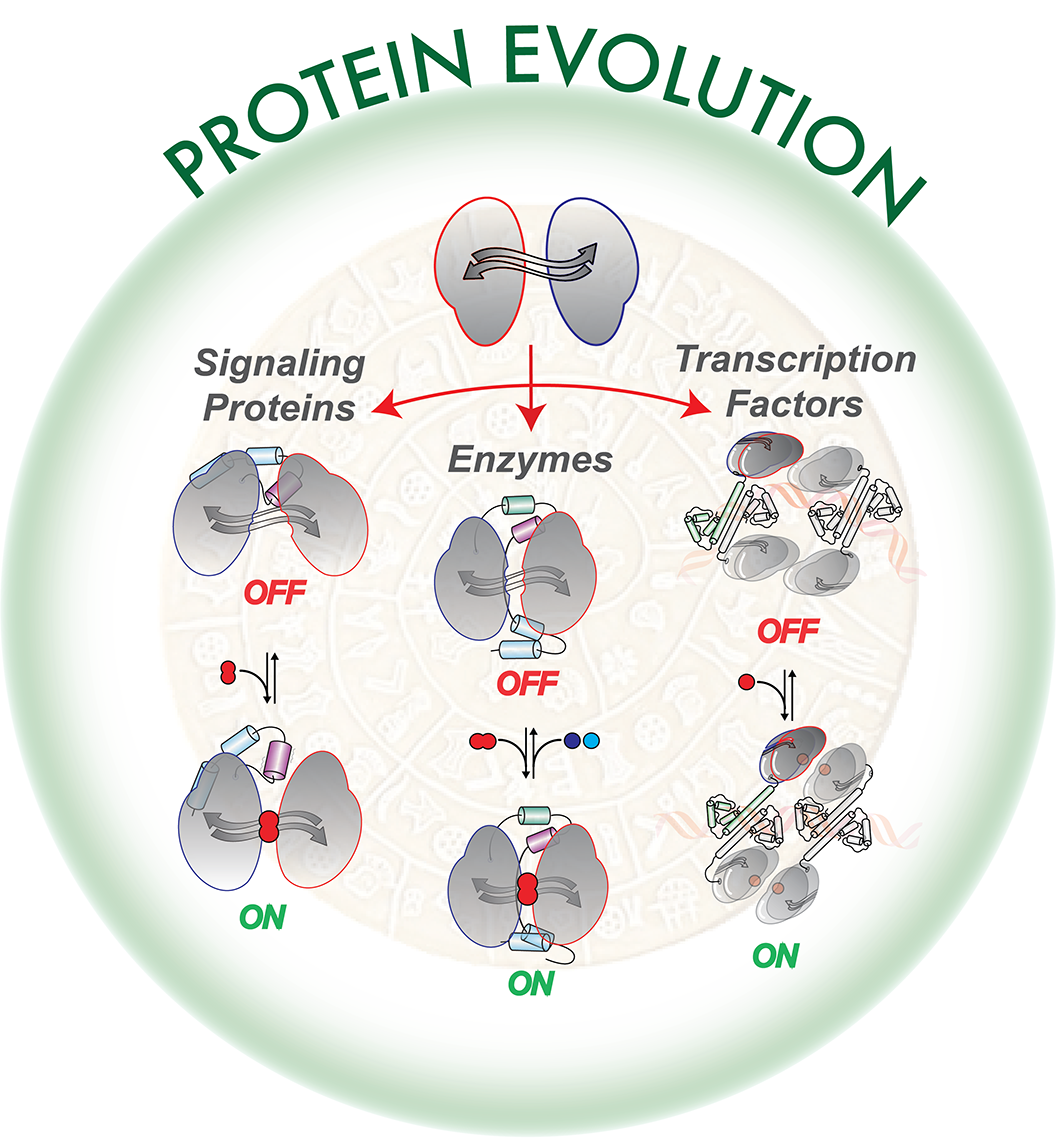
Proteins are responsible nearly for all processes occurring within cells. Thus, protein malfunction represents the molecular etiology of diseases and death. The activity of such biomolecules relies on their capacity to vary their shape or structure over time. Such variations are finely-regulated through their interaction with small molecules and/or other biopolymers, and termed Structural Dynamics. Research conducted at the IMBB has shown that Structural Dynamics have been essential for the proteins to adapt to the continuously fluctuating chemical environment over the last 3.5 billion of years on earth.
For this purpose, structural, evolutionary and biophysical analysis was performed on about 600 extant proteins with a conserved and ancestral structural core encountered throughout the tree of life. Modifications of this structural core, enabled distinct Structural Dynamics which diversified its fate during evolution, giving rise to protein acting as signaling, transcription factors, enzymes, or conferring transport-related functions. These findings are likely to be applicable to a large number of protein structures.

Dr. Giorgos Gouridis and his colleagues Babis Pozidis and Dr. Yusran Muthahari
This research is the result of an international collaboration between the Dynamic Structural Biology Team led by Dr. Giorgos Gouridis and his colleagues Babis Pozidis and Dr. Yusran Muthahari with the Ludwig Maximilian University of Munich (Germany), and the contribution of researchers from the Universities of Groningen (Netherlands) and Leuven (Belgium).
Link to the publication: https://www.pnas.org/content/118/49/e2026165118
More info:
Giorgos Gouridis
Researcher, IMBB-FORTH
Email: g.gouridis@imbb.forth.gr | Tel.: +30-2810-391056
https://www.imbb.forth.gr/en/research-en/item/5040-giorgos-gouridis